MONDAY, 21 DECEMBER 2015
Dengue fever is a virus-driven disease causing a worldwide burden. The WHO estimates that 500,000 infected people are hospitalised each year due to severe dengue symptoms and 2.5% of these subsequently die. The virus is transmitted among humans by a mosquito bite (Aedes spp.). While some efforts are put into developing therapies that alleviate the negative symptoms of the disease in patients, scientists are focusing on new approaches to reduce the infection in mosquitos and therefore prevent its transmission to humans.One way of reducing virus infection in mosquitos is targeting mosquito genes that regulate viral load. A team lead by Professor Tonya Colpitts and funded by the National Institutes of Health recently identified a candidate gene for this job. In this new study, the authors focused on understanding the role of a gene that codes for a cysteine-rich venom protein (named CRVP379). Their study found a strong association between the level of viral infection in mosquitos and the protein expression at both global and tissue-specific levels in in vitro and in vivo experiments. Further experimental manipulations showed that blocking CRVP379 protein with either RNAi, which they found interacts with viral receptor prohibitin, could be used to block mosquito infection.
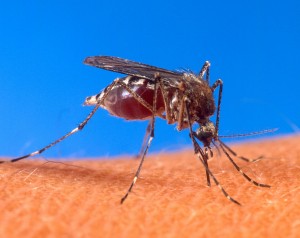 One way of reducing virus infection in mosquitos is targeting mosquito genes
One way of reducing virus infection in mosquitos is targeting mosquito genesDOI:10.1371/journal.ppat.1005202
Written by Anaid Diaz.
