FRIDAY, 25 JANUARY 2013
Inside our cells we have the result of an unexpected event that allowed us to evolve from a single cell to the complex multicellular organisms we are today. This tiny life changing structure or organelle is the mitochondrion, and is often described as the power plant of the cell as it is responsible for providing the energy a cell needs to carry out a number of important functions.Scientists believe that mitochondria evolved from a free-living bacterium that became engulfed and sustained by a larger single-celled organism in a process known as endosymbiosis.
In return for food and shelter the smaller organism provided the larger cell with energy allowing the cell to become more complex over many generations until it became the cells we have today. Over time mitochondria lost many of their genes and became dependent on the host for its replication. Now just an organelle, the mitochondria retains a set of 37 genes that help it to operate.
Mutations in mitochondrial genes or in nuclear genes that encode mitochondrial proteins can lead to disease and even death. Unlike most genetic diseases where errors in the nuclear genes can be inherited from either parent, defects in mitochondrial DNA are strictly inherited from the mother only.
Mitochondria are present in the unfertilized egg and once the egg is fertilized the embryo divides and replicates them, resulting in almost every cell in the body having mutated and defective mitochondria. Mitochondrial diseases – of which around 50 are known – affect one in every 6,500 people. Although many sufferers have mild or no symptoms, defective mitochondria can cause severe health problems affecting a number of organs, particularly the organs that require the most energy like the brain, heart and muscles. Despite ongoing research at present no treatment is available and, even in the best-case scenarios, clinicians can only manage the symptoms that arise.
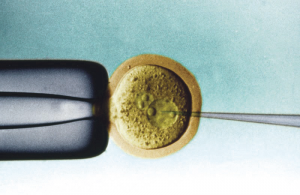
Scientists, both in the United States and the United Kingdom, have been working on new techniques based on In Vitro Fertilization (IVF) technology to prevent mitochondrial diseases that are caused by faulty mitochondrial DNA (mtDNA).
The first technique developed, pro-nuclear transfer (PNT), is performed immediately after fertilization. Scientists remove the pronucleus, which is derived from the nuclei of the egg and sperm, from an embryo with unhealthy mitochondria and transfer it into a donated embryo with healthy mitochondria that has had its pronucleus removed. This new embryo contains nuclear DNA from the intended father and mother and healthy mtDNA from a third donor. A second technique, Maternal Spindle Transfer (MST), involves the transfer of DNA from the mothers egg to a enucleated egg with healthy mitochondria before fertilization. MST differs principally from PNT in that eggs, rather than embryos, are destroyed.
Mitochondrial replacement techniques hold great promise for women who carry genes that cause mitochondrial disease, but who want to have children. They eliminate the risk of genetic disease being passed down to future generations. Although these techniques have had success in the lab, with healthy “three-genome” primates being born, there approval for use in the clinic is still a long way off. Mitochondrial replacement would result in a modified embryo creating human life, a medical first, and this raises a number of ethical questions that need to be addressed before the technology can be routinely used.
The main issue arising from mitochondrial replacement is that the resulting child will have DNA from the mother and father which will give it its characteristics, but it will also have a tiny amount of mitochondrial DNA from a third party female donor. Even though this foreign mtDNA would comprise only 0.2 per cent of the child’s DNA, this use of extra genetic material has lead to sensationalist headlines about “three-parent babies”. This is a misleading term because it suggests the resulting child would exhibit external characteristics of the donor, something that is not entirely true. Even so, the child would have mtDNA from a third person, and opponents of MST and PNT worry that this may impact on the child’s sense of identity and alter society’s perception of parenthood.
Crucially this is the first genetic modification of a human to be incorporated into the germ line, the DNA that passes onto future generations, something that is currently banned in the UK. There is also the concern that changing the heritable characteristics of a person may lead to a “slippery slope” where it may be seen to be morally just to modify embryos in more trivial ways in the future, for example modifying the genes which give us our characteristics such as height and hair colour.
Questions have also arisen about the safety of the technique, not only for the baby who results from the egg, but also for the child’s descendants, as opponents of the procedure fear that any biological faults introduced by the technology may not have consequences for a number of generations.
A number of groups, including the UK’s leading centre for Bioethics; the Nuffield council for Bioethics, believe the treatment to be ethical if it is found to be safe and effective, something which the scientists at Newcastle University carrying out the research hope to show in the next five years. There will inevitably be risks if and when the procedure is tried in humans, but these must be weighed against the risks associated with mitochondrial disease.
As with any new technology dealing with the creation of life and genetic modification this is an emotive issue and the regulatory body the Human Fertilization and Embryology Authority (HFEA) has held a public consultation, the results of which will be available in early 2013, to gage public attitudes prior to a parliamentary vote on changing the law to allow three genome embryos to be created.
The debate on whether MST and/or PNT should be allowed is centred in the United Kingdom as no other country is as close in being able to offer this technology to patients. Other countries conducting research in mitochondrial replacement, such as the US, have strict laws and funding restrictions that govern the use of human embryos.
Although many feel that creating genetically modified children is a line we should not cross, for many parents affected by mitochondrial disorder, this may be their only chance to have a child free or a potentially fatal genetic disease. Whatever the politicians decide in the parliamentary vote, the debate on mitochondrial replacement will continue with advances in reproductive technology.
Nicola Love is a PhD student in the Department of Physiology, Development and Neuroscience